42 reaction products worksheet answers
Reaction Time: The Ruler Drop Test | Science project - Education The time it takes from when your eye first notices the ball to when your arm reaches up to catch it is an example of reaction time. Even though stimuli—or changes in your environment that you react to—travel very quickly along your nervous system as messages, your body doesn’t react instantly. Many athletes spend hours practicing to ... Cathode Ray Experiment by JJ.Thomson (CRT) - Explanation Cathode Ray Tube - The Cathode Ray Experiment by J.J.Thomson helped to discover electrons. Cathode ray tube is the heart of the oscilloscope and it generates the electron bean, accelerates the beam and deflects the beam. Visit BYJUS to learn more about it.
Physical and Chemical Changes Worksheet combust making new products and energy. Part C: True (T) or False (F) 1. F Changing the size and shapes of pieces of wood would be a chemical change. 2. F In a physical change, the makeup of matter is changed. 3. T Evaporation occurs when liquid water changes into a gas. 4. T Evaporation is a physical change. 5. F Burning wood is a physical ...
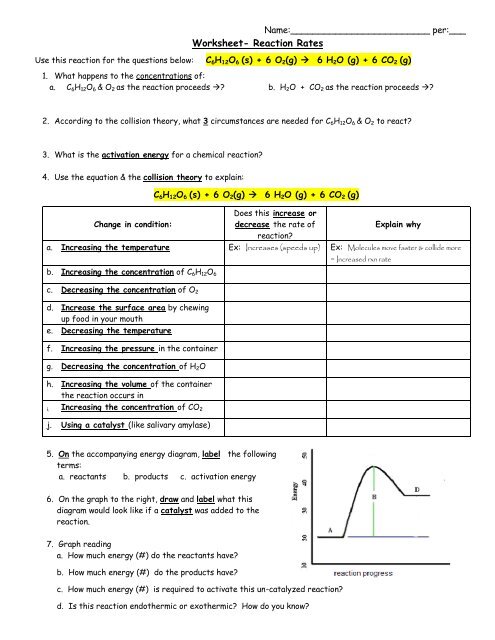
Reaction products worksheet answers
2.5: Reaction Rate - Chemistry LibreTexts 17/06/2022 · Definition of Reaction Rate. The Reaction Rate for a given chemical reaction is the measure of the change in concentration of the reactants or the change in concentration of the products per unit time. The speed of a chemical reaction may be defined as the change in concentration of a substance divided by the time interval during which this change is observed: THE PROPERTIES AND STRUCTURE OF MATTER • Chemical reaction consists of reactants and products reactants – starting substances products – substances which are form/produced . Law of Conservation of Mass • The law of the conservation of mass applies to chemical reactions • Mass is neither created nor destroyed during a chemical reaction • Mass is conserved! • Mass reactants = Mass products. Classify … Similarities Between Photosynthesis And Cellular Respiration Cellular respiration takes a glucose molecule and combines it with oxygen; the result is energy in the form of ATP, along with carbon dioxide and water as waste products. Photosynthesis takes ...
Reaction products worksheet answers. Gibbs Free Energy - Purdue University The entropy term is therefore subtracted from the enthalpy term when calculating G o for a reaction.. Because of the way the free energy of the system is defined, G o is negative for any reaction for which H o is negative and S o is positive. G o is therefore negative for any reaction that is favored by both the enthalpy and entropy terms. We can therefore conclude that any … Difference Between Physical and Chemical Change - BYJUS During a chemical reaction, absorption and evolution of energy take place. Some examples of physical change are freezing of water, melting of wax, boiling of water, etc. A few examples of chemical change are digestion of food, burning of coal, rusting, etc. Generally, physical changes do not involve the production of energy. Chemical changes usually involve the production of … KINETICS Practice Problems and Solutions Identify the location of reactants, intermediate(s), activated complex(es), and products. 9. A first-order reaction is 38.5% complete in 480 s. a. Calculate the value of the rate constant. b. What is the value of the half-life. c. How long will it take for the reaction to reach 95% completion. 10. The rate of the reaction NO 2 + CO → NO + CO 2 depends only on the concentration of nitrogen ... 100 Balancing Chemical Equations Worksheets with Answers 02/10/2019 · This signifies a reaction which is irreversible or is unchangeable after a certain stage. However, in certain situations, the reactions occur at equilibrium. This means that reaction at any forward rate results in a reverse reaction. In such situations, the arrow used is two-sided, i.e. facing towards the reactants and the products.
Similarities Between Photosynthesis And Cellular Respiration Cellular respiration takes a glucose molecule and combines it with oxygen; the result is energy in the form of ATP, along with carbon dioxide and water as waste products. Photosynthesis takes ... THE PROPERTIES AND STRUCTURE OF MATTER • Chemical reaction consists of reactants and products reactants – starting substances products – substances which are form/produced . Law of Conservation of Mass • The law of the conservation of mass applies to chemical reactions • Mass is neither created nor destroyed during a chemical reaction • Mass is conserved! • Mass reactants = Mass products. Classify … 2.5: Reaction Rate - Chemistry LibreTexts 17/06/2022 · Definition of Reaction Rate. The Reaction Rate for a given chemical reaction is the measure of the change in concentration of the reactants or the change in concentration of the products per unit time. The speed of a chemical reaction may be defined as the change in concentration of a substance divided by the time interval during which this change is observed:





![49 Balancing Chemical Equations Worksheets [with Answers]](https://templatelab.com/wp-content/uploads/2017/01/balancing-equations-32.jpg)









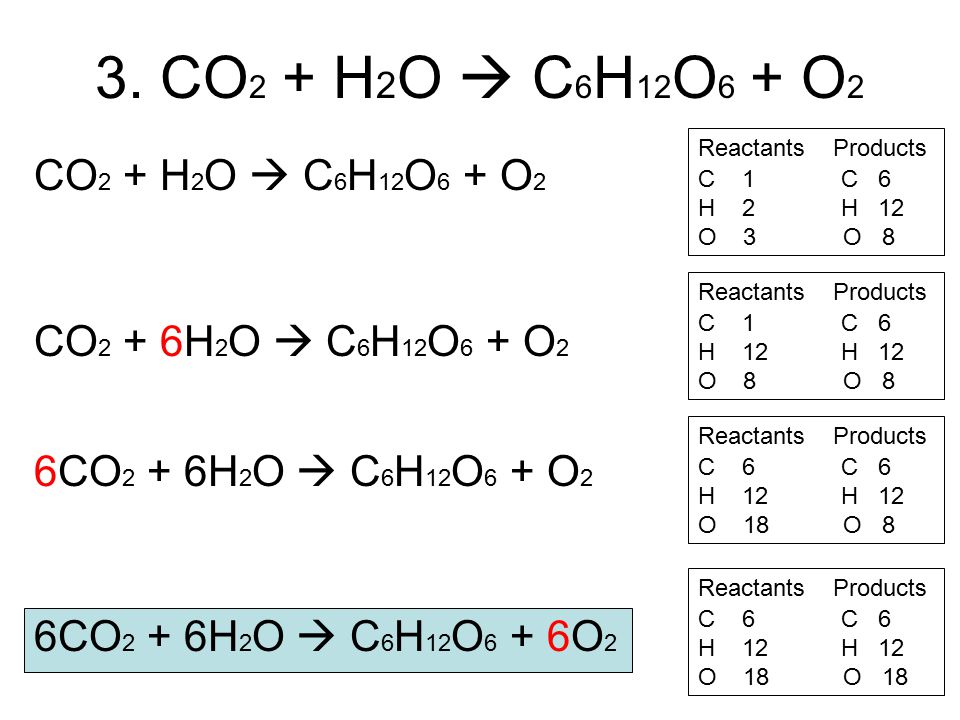





![49 Balancing Chemical Equations Worksheets [with Answers]](https://templatelab.com/wp-content/uploads/2017/01/balancing-equations-10.jpg)







0 Response to "42 reaction products worksheet answers"
Post a Comment